Serotonin, or rather its deficiency, frequently steals the spotlight in conversations regarding depression. Initially discovered as a component of serum in 1948 to regulate vasoconstriction (serotonin = serum + tone), the role of serotonin in depressive disorders wasn't implied until a few years later. Since then, much effort has been dedicated by scientists and clinicians alike to understand the wondrous complexity of the seemingly inscrutable code that is serotonin neurotransmission. In fact, some of the major breakthroughs in psychopharmacology happened with serotonin in mind – the discovery of selective serotonin reuptake inhibitors (SSRIs) prominently expanded the therapeutic toolbox for mental health practitioners.
The appearance of SSRIs, however, was just the beginning of the arduous journey to understand and treat the intricate disorder that is depression. Providing valuable relief for some by offering mood improvement and stability, the treatment of depression has fallen short of optimal for others [1]. As brilliant research minds continue to unravel the mysteries of depression, what is becoming clearer is that depression is prolific in complexity, painting a picture of inappropriate entanglement of neural and somatic pathways as they respond to a variety of insults. It is this complexity that makes it difficult to understand where and how the intrinsic program has gone off the rails, leaving practitioners without significant leverage in the plight to increase a patient’s emotional reserve.
Recent explosions of interest in brain studies are actively driving research in depression to the next league. But the enormity of the task to uncover relevant underpinnings of depression still remains overwhelming. And that probably has to do with the fact that at its most fundamental level, depression is excruciatingly heterogeneous. An astounding number of factors could be responsible for the derailed mood infrastructure - dysregulation of inflammatory, metabolic, neuroendocrine, growth factor and of course, neurotransmitter systems, just to name a few [1].
Time for Pie
To describe this in simpler terms, let’s think of depression for a moment as a pumpkin pie. Each slice within the pie represents a particular system which, when dysregulated, contributes to the pathology of the disorder. In this over-simplified model of depression, neurotransmitters are just one slice of the pie, and within that slice, there are even smaller slices that could be any or all of the following – serotonin, dopamine, norepinephrine, glutamate - you name it! What does this mean for serotonin in particular? Well, serotonin happens to be the piece that gets talked about a lot because historically it’s the most-studied piece. In the monoamine theory of depression, serotonin is a prominent player. But unlike this two-dimensional model of pie slices that are nestled comfortably in their own pie framework, the real “pie pieces” are dynamic and frequently affect how and what the other “pie pieces” are doing.
This blog is going to focus on a small but significant aspect of serotonin biology that pertains to depression. If prominent serotonin lowering is to blame for the depressive symptoms in a subset of patients, then what are some of the endocrine factors that could be driving and perpetuating this dearth of neurochemicals? The mechanisms behind hormonal regulation of serotonin levels are key to our understanding of the patient’s mood pathology.

The "Housekeeping" Functions of Serotonin
A small molecule with such profound impact, serotonin fulfills an impressive number of critical roles throughout the body – above and below the neck. Serotonin is our body’s housekeeping chemical –it promotes feelings of well-being, hence the nickname “happiness molecule”. Serotonin also arms us against adversity, providing us with resilience. And it does so much more! Serotonin regulates appetite, temperature, energy balance, platelet coagulation, bone remodeling, sleep cycles, emesis, the inflammatory response and sexual behavior, just to name a few [2]. To make all these processes happen, serotonin works in a dynamic equilibrium relying on communication from other molecules – estradiol, testosterone, cortisol, vitamin D and many others that help shape its molecular behavior. In this context, hormones play a very important role in modulating serotonin signaling.
To properly address insufficient serotonin levels, we need to understand how the body makes this neuro-chemical.
A Word About How Serotonin is Made...
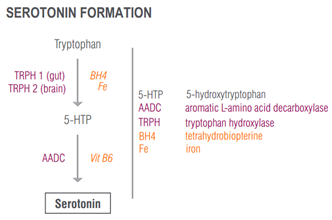
To synthesize serotonin in the brain and the rest of the body, humans start with tryptophan, an amino acid found in foods like meats and fish, dairy and tofu, nuts and seeds [3]. Conversion of tryptophan to serotonin is a two-step process. First, tryptophan is converted to its downstream metabolite, 5-HTP. Tryptophan hydroxylase (TRPH) is the enzyme in charge of this process – type 1 does the job in the gut (where upwards of 90% of all serotonin is manufactured in the body), and type 2 reigns in the brain. Sufficient biopterin and iron are required to facilitate the activity of TRPH and get over the hurdle of this rate-limiting step [4] [5].
Once 5-HTP is formed, the path to serotonin is smooth sailing. Sure, there’s still a need for vitamin B6 in order for the reaction to proceed, but typically 5-HTP to serotonin is a straightforward biochemical transaction. This is why for serotonin-deficient individuals, 5-HTP supplementation can be an effective therapeutic strategy to elevate mood either as a stand-alone treatment [6] or in conjunction with SSRIs [7] [8].
... and How Serotonin is Broken Down
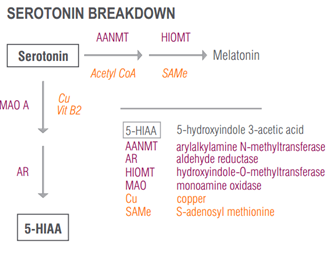
Serotonin is broken down to its inactive metabolite 5-HIAA via a 2-step process, one of which is governed by monoamine oxidase A (MAO A). (Note that not all serotonin metabolites are inactive; for example, melatonin is derived from serotonin to regulate sleep). There has to be enough MAO A to assist with keeping serotonin levels “just right” – too much MAO A activity and the serotonin pool will be zealously depleted, leaving the individual vulnerable to afflictions such as insomnia, excessive worry, and perhaps even depressive mood. Not enough MAO A activity, and serotonin levels may build up, potentially contributing to a multifaceted array of aggressive and impulsive behaviors [9].
To get a glimpse at MAO A activity in a patient, assaying serotonin and 5-HIAA together is imperative. The levels of both species – the neurotransmitter and its metabolite – are analyzed together to make the most informed therapeutic decision. Unlike a straight-lined seesaw, the levels of serotonin and 5HIAA don’t always change in a linear fashion in relation to each other and utilizing that discrepancy can be of important clinical value.
To summarize, TRPH is important for serotonin biosynthesis, and MAO A is important for its metabolism. These steps in the biosynthesis and breakdown of serotonin are precisely the check-points where hormones can step in and acutely regulate serotonin levels. Below is a summary of how different hormones modulate serotonin levels through biosynthesis and breakdown.
The Nurturing Nature of Estradiol Towards Serotonin
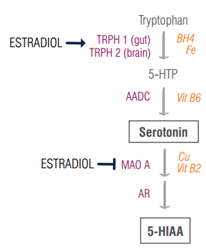
Estradiol is especially nurturing towards serotonin – it stimulates TRPH expression to ensure that enough serotonin is made and suppresses MAO A levels to prolong the longevity of the neurotransmitter. This relationship between estradiol and serotonin is prominently featured in perimenopause, when estradiol levels eventually plummet, leaving serotonin down in the dumps as well. This is one of the reasons why mood disorders are common in menopause – when estradiol goes away, so can serotonin [10].
Hormone replacement therapy can effectively manage existing depression in menopause as it aims to restore estradiol to pre-menopausal physiological levels [11]. Moreover, a recent double-blind, placebo-controlled randomized trial published in JAMA Psychiatry provides some of the first evidence that depressive symptoms can actually be prevented with hormone replacement therapy, thus protecting women from developing mood disorders as a consequence of extreme estradiol fluctuations/withdrawal [12]. SSRIs can also help take the edge off low mood, although some women may be unwilling to comply because of a smothered libido (which is already low because of low estradiol).
Menopause is not the only time when the estradiol/serotonin connection becomes critical with respect to mood. In pregnancy, estradiol levels increase by upward of 30-fold compared to levels prior to conception. Shortly after birth, with the loss of the placenta, estradiol is rapidly cleared to usher in the lactation period, leaving some women particularly vulnerable to developing postpartum depression. In the United States alone, approximately 1 in 9 women experience depressive symptoms after giving birth [13]. Persistent depressive symptoms that develop after having a baby, also called postpartum depression, range in prevalence – from a low of 8% to a high of 20% in some states with an average of 13% across the country [13].
So now for that connection with serotonin. A recent study utilized imaging techniques to look at MAO A levels in the postpartum period. Women with postpartum depression had elevated MAO A density in the brain areas responsible for emotion compared to asymptomatic women in the postpartum period [14].(Note: the higher the levels of MAO A, the lower the levels of monoamine neurotransmitters, including serotonin. Elevated MAO A activity has been reported in other types of depression [15].) As estradiol drops rapidly in the first few days postpartum, there is a temporary rise in MAO A levels and activity. But for some women, MAO A levels fail to return to normal, and remain elevated, all the while hastily burning through available serotonin. Without adequate normalization, these elevated MAO A levels, compounded by the challenges that motherhood brings, renders some women overtly prone to developing postpartum mood disorders.
In summary, the serotonin system learns to rely heavily on estradiol from the onset of puberty and beyond. In circumstances when estradiol levels decrease profoundly, such as in postpartum when estradiol goes from high to physiological, or in menopause when estradiol drops from normal to essentially non-existent, the serotonin system can struggle to adjust. Loss of equilibrium in the serotonin system can then manifest as mood disorders.
The Caveat of Serotonin Lowering by Testosterone
First isolated 80-some years ago, testosterone’s influence on the brain is finally getting much needed research attention rather than being largely a matter of creative speculation.
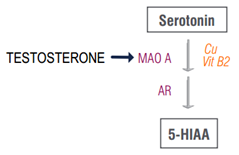
The relationship between testosterone and serotonin has not been fleshed out as well as it has for estradiol. However, experimental evidence suggests that testosterone does the opposite of estradiol – it potentiates MAO expression. In the presence of an aromatase inhibitor to prevent testosterone metabolism to estradiol, testosterone actively elevates MAO A levels, consequently lowering precursor neurotransmitters (serotonin) and elevating metabolites (5-HIAA) [16].
But this is where the old-fashioned dualism of the line dividing androgens and estrogens becomes blurry and not well-defined. Many of testosterone’s effects on the brain are paradoxically estrogenic in nature. This is because the rapid generation of estrogen from its yangian counterpart testosterone in the brain, an organ which is rich in aromatase, results in suppression of MAO A.
Nature abhors absolutes and this testosterone-estradiol push-pull in regulating one of the most prominent monoamine metabolizing enzymes is a prime example of how it keeps a balance in a system as delicate as the brain.
An Unexpected Kinship Between Cortisol and Serotonin
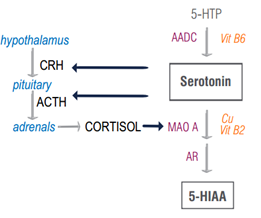
Stress is one of the primary risk factors for developing mood pathologies. When threats are chronic, unrelenting and intense in nature, vulnerable individuals respond by the consequent hyperactivation of the HPA axis resulting in persistently elevated cortisol levels. The stress cascade becomes a wrecking crew, spearheaded by cortisol, that targets neurotransmitters with utmost ferocity. Studies show that a subset of patients suffering from depressive disorders display a hyper-aroused HPA axis with higher levels of cortisol compared to individuals who are not depressed [17] [18]. (Note: although elevated cortisol is observed in some depressed patients, it is not a defining feature for all types of depression.) And the functional consequence of elevated cortisol may just be diminished serotonin levels, because cortisol is yet another member of the neuroendocrine family with MAO A-potentiating capabilities [19]. Very exciting!
Yet the plot thickens even more when serotonin emerges not merely as an innocent passive bystander, simply receiving inputs from a devouring MAO A governed by an elevated glucocorticoid tone, but as an active, prominent modulator of the HPA axis in and of itself. The serotonin system (serotonin + serotonin receptors) acts directly in the hypothalamus to stimulate the production of CRH, corticotropin-releasing hormone, an essental part of the cortisol-activation cascade [19]. In fact, 5-HTP administration triggers an impressive increase in salivary cortisol levels compared to placebo [20]. This spike in salivary cortisol in response to 5-HTP may be tolerated, without adverse symptomatology, by many individuals. However, in patients with panic disorder, cortisol levels spike even higher than in controls [20]. Thus, the subsidiary effect of increased agitation with 5-HTP supplements in sensitive individuals may be explained by a rapid rise in cortisol.
The Low Down on Vitamin D and Serotonin
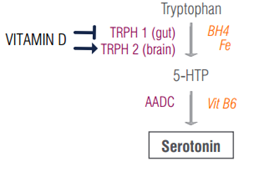
Interest in vitamin D has been growing exponentially as large swaths of the population are not getting enough of this "sunshine vitamin." Biologically a hormone, produced in the skin in response to sunlight, vitamin D is important for mood health. Disappearance of vitamin D has been linked to mood disorders, such as seasonal affective disorder, mania [21], psychosis [22], and depression [23].
With respect to serotonin, adequate vitamin D levels are essential for appropriate serotonin biosynthesis. Like estradiol, vitamin D potentiates the expression of neuronal TRPH to stimulate the appropriate production of serotonin in the brain [24] [25]. But remember how there are two types of TRPH and type 1 is present in the gut to regulate the majority of the body’s serotonin production? Well, vitamin D inhibits the expression of TRPH 1 to suppress serotonin biosynthesis [24].
What does this mean for a patient testing their urinary serotonin who is concurrently deficient in vitamin D? Without sufficient vitamin D, this individual may be suffering from low brain serotonin (not enough TRPH 2 stimulation) all the while having high serotonin production in the gut (not enough TRPH 1 suppression). This can explain why a patient who checks depression on the symptom portion of the ZRT requisition form may have a high serotonin result.
In Conclusion
As with all biochemical systems, none can be viewed in isolation. Hormonal changes are as involved with neurotransmitter systems as they are with the other physiological systems they impact. A better understanding of the interplay between these systems can go a long way to helping practitioners view their patients with depression by getting to the source of the imbalance rather than treating only the end result.
Related Resources
- Blog: Examining the "Warrior Gene" - MAOA & Aggression
- Blog: Shorter Days - A SAD Pattern
- Blog: Clinical Pearls - Getting the Most Out of Your Neurotransmitter Test
References
[1] Strawbridge, R., A.H. Young, and A.J. Cleare, Biomarkers for depression: recent insights, current challenges and future prospects. Neuropsychiatr Dis Treat, 2017. 13: p. 1245-1262.
[2] Shajib, M.S. and W.I. Khan, The role of serotonin and its receptors in activation of immune responses and inflammation. Acta Physiol (Oxf), 3/2015. 213(3): p. 561-574.
[3] Strasser, B., J.M. Gostner, and D. Fuchs, Mood, food, and cognition: role of tryptophan and serotonin. Curr. Opin. Clin Nutr Metab Care, 1/2016. 19(1): p. 55-61.
[4] Hasegawa, H. and K. Nakamura, CHAPTER 2.3 - Tryptophan Hydroxylase and Serotonin Synthesis Regulation, in Handbook of Behavioral Neuroscience, C.P. Müller and B.L. Jacobs, Editors. 2010, Elsevier. p. 183-202.
[5] Kim, J. and M. Wessling-Resnick, Iron and mechanisms of emotional behavior. J Nutr Biochem, 2014. 25(11): p. 1101-1107.
[6] Boadle-Biber, M.C., Regulation of serotonin synthesis. Prog Biophys Mol Biol, 1993. 60(1): p. 1-15.
[7] Kious, B.M., et al., An Open-Label Pilot Study of Combined Augmentation With Creatine Monohydrate and 5-Hydroxytryptophan for Selective Serotonin Reuptake Inhibitor- or Serotonin-Norepinephrine Reuptake Inhibitor-Resistant Depression in Adult Women. J Clin Psychopharmacol, 2017. 37(5): p. 578-583.
[8] Jacobsen, J.P.R., et al., Adjunctive 5-Hydroxytryptophan Slow-Release for Treatment-Resistant Depression: Clinical and Preclinical Rationale. Trends Pharmacol Sci, 2016. 37(11): p. 933-944.
[9] Godar, S.C., et al., The role of monoamine oxidase A in aggression: Current translational developments and future challenges. Prog. Neuropsychopharmacol. Biol. Psychiatry, 8/1/2016. 69: p. 90-100.
[10] Bromberger, J.T. and H.M. Kravitz, Mood and menopause: findings from the Study of Women's Health Across the Nation (SWAN) over 10 years. Obstet. Gynecol. Clin. North Am, 9/2011. 38(3): p. 609-625.
[11] Fischer, B., C. Gleason, and S. Asthana, Effects of hormone therapy on cognition and mood. Fertil. Steril, 4/2014. 101(4): p. 898-904.
[12] Gordon, J.L., et al., Efficacy of transdermal estradiol and micronized progesterone in the prevention of depressive symptoms in the menopause transition: A randomized clinical trial. JAMA Psychiatry, 2018.
[13] Ko, J.Y., et al., Trends in Postpartum Depressive Symptoms - 27 States, 2004, 2008, and 2012. MMWR Morb Mortal Wkly Rep, 2017. 66(6): p. 153-158.
[14] Sacher, J., et al., Relationship of monoamine oxidase-A distribution volume to postpartum depression and postpartum crying. Neuropsychopharmacology, 2015. 40(2): p. 429-35.
[15] Meyer, J.H., et al., Elevated monoamine oxidase a levels in the brain: an explanation for the monoamine imbalance of major depression. Arch. Gen. Psychiatry, 11/2006. 63(11): p. 1209-1216.
[16] Bethea, C.L., et al., Androgen metabolites impact CSF amines and axonal serotonin via MAO-A and -B in male macaques. Neuroscience, 2015. 301: p. 576-89.
[17] Gupta, S., et al., Evaluation of Endocrine Parameters as Predictor of Major Depressive Disorder. Indian J Psychol Med, 2017. 39(6): p. 766-769.
[18] Duval, F., et al., Interaction between the serotonergic system and HPA and HPT axes in patients with major depression: implications for pathogenesis of suicidal behavior. Dialogues Clin Neurosci, 2002. 4(4): p. 417.
[19] Higuchi, Y., T. Soga, and I.S. Parhar, Regulatory Pathways of Monoamine Oxidase A during Social Stress. Front Neurosci, 2017. 11: p. 604.
[20] Schruers, K., et al., L-5-hydroxytryptophan induced increase in salivary cortisol in panic disorder patients and healthy volunteers. Psychopharmacology (Berl), 2002. 161(4): p. 365-9.
[21] Altunsoy, N., et al., Exploring the relationship between vitamin D and mania: correlations between serum vitamin D levels and disease activity. Nord J Psychiatry, 2018: p. 1-5.
[22] Hedelin, M., et al., Dietary intake of fish, omega-3, omega-6 polyunsaturated fatty acids and vitamin D and the prevalence of psychotic-like symptoms in a cohort of 33 000 women from the general population. BMC Psychiatry, 2010. 10: p. 38-38.
[23] Bahrami, A., et al., High Dose Vitamin D Supplementation Is Associated With a Reduction in Depression Score Among Adolescent Girls: A Nine-Week Follow-Up Study. J Diet Suppl, 2017: p. 1-10.
[24] Patrick, R.P. and B.N. Ames, Vitamin D hormone regulates serotonin synthesis. Part 1: relevance for autism. FASEB. J, 6/2014. 28(6): p. 2398-2413.
[25] Patrick, R.P. and B.N. Ames, Vitamin D and the omega-3 fatty acids control serotonin synthesis and action, part 2: relevance for ADHD, bipolar disorder, schizophrenia, and impulsive behavior. Faseb J, 6/2015. 29(6): p. 2207-2222.