
Attention deficit hyperactivity disorder (ADHD) is the most common neurodevelopmental disorder in children; however, boys are diagnosed two to nine times more often than girls are (1). Girls do have ADHD, but it often goes unnoticed because it can present much differently than it does in boys. Girls tend to be quiet and inattentive whereas boys tend to be active and disruptive. Children with ADHD can present with symptoms of inattention, hyperactivity/impulsivity, or all of the above, although symptoms may also shift over time (https://www.cdc.gov/ncbddd/adhd/diagnosis.html).
The presentation of ADHD in girls is typically not disruptive to classroom activity, so it may go unnoticed until later in life when coexisting conditions arise either as a consequence of undiagnosed ADHD, or as independent diagnoses. ADHD can present as depression, anxiety, bipolar disorder, issues with self-esteem, underachievement, isolation, and difficulty forming friendships (2). The presence of coexisting disorders tends to reduce the likelihood that girls and young women will be diagnosed with ADHD as many providers are quick to attribute their symptoms of inattentiveness or impulsivity to depression or bipolar disorder (1).
When to suspect ADHD in girls and women
Before diagnosing other disorders in girls who are having issues with self-esteem, difficulty in school or at work, or problems with friendships and other relationships, it is important to determine if there are other family members with ADHD. There is no simple etiology because both genetic and environmental factors can increase the likelihood for ADHD; however, having a first-degree relative with ADHD is associated with a two- to eight-fold increased likelihood regardless of gender (2). Studies in twins indicate that 75%–90% of ADHD is caused by genetic factors. If one person in a family is diagnosed with ADHD there is a 25%–35% probability that another family member also has ADHD. Approximately 50% of parents who have ADHD will have a child with the disorder (3).
Symptoms of ADHD in girls may present as daydreaming, inattentiveness, anxiety, and shyness. They may also have a more impulsive-type presentation expressed as excessive talking, nervousness, risk-taking behavior, and a tendency to be domineering. During adolescence and up to 25 years of age, frontal lobe development related to skills in executive function are emerging and developing. In those with ADHD, this development is delayed, and the lack of skills promotes additional anxiety and dysfunctional behavior furthering issues with self-esteem (2).
For most people, ADHD is not something that you outgrow so diagnosis in adulthood is not uncommon. The mean age of diagnosis in women who have not been diagnosed in childhood is 36 to 38 years of age. The diagnosis often occurs because of a diagnosis in their own children. Prior to a diagnosis of ADHD, these women may have been diagnosed with a mood or anxiety disorder (2). Though these conditions can coexist with ADHD, they may also be a misdiagnosis because ADHD was never considered as the primary cause of their symptoms.
Coping with ADHD
Women who are not aware that they have ADHD may exist with the constant feeling that they can’t seem to measure up to their peers. To make up for a feeling of lack, some women with ADHD can go to the extreme of perfectionism and diligence in certain areas of their lives to attain a sense of control over the internal overwhelm that seems to define their existence. Many women do not present with the physical hyperactivity of ADHD, but that hyperactivity is turned inward and manifests as a racing mind and a sense of overwhelm (1).
Through maturity and necessity, women learn various coping mechanisms and strategies that help them to manage what may feel like a level of internal chaos. Life often becomes overwhelming for young and middle-aged women as they attempt to manage life, work, children, and social expectations (1). Most of these women are very intelligent and develop strategies to cope with their own personal idiosyncrasies; however, the need to implement these coping mechanisms can be exhausting as it adds an extra layer of complexity to an already complex life.
Hormones and ADHD
Specific for women with ADHD are the hormonal fluctuations that accompany puberty, monthly menstrual cycles, pregnancy, perimenopause, and menopause. Hormonal fluctuations can be more extreme in women with ADHD as there is already a tendency towards emotional dysregulation. Depression, premenstrual syndrome and premenstrual dysphoric disorder tend to be more common in women who have ADHD. Symptoms of ADHD can be better during the late follicular and ovulatory phases when estrogen is rising and worse during the luteal phase when progesterone is rising and potentially decreasing the beneficial effects of estrogen on the brain (4). Overall, hormonal fluctuations and transitional periods of life can influence the symptoms and presentation of ADHD in women (2).
Estradiol has complex and widespread effects on the nervous system. The aging process that occurs in the nervous system is closely related to the aging process in the endocrine system. The perimenopausal phase of a woman’s life is often characterized by symptoms such as headaches, sleep disturbance, mood fluctuations, anxiety, depression, and impaired cognitive function. When the hormonal balance is disrupted, women are at greater risk of developing neurocognitive dysfunction (5).
A diagnosis of ADHD may occur at the time of menopause because the severity of hormonal decline unmasks the underlying diagnosis. Correspondingly, the decline in executive function may simply appear similar to the symptoms of ADHD because of the effects of declining hormones on neurotransmitter levels and cognitive function.
Executive function and menopause
Executive function is a set of mental skills that includes working memory, cognitive flexibility, and self-control. We use these skills to learn, focus, and manage everyday life. Issues with executive function make it difficult to focus, plan, organize, prioritize, finish tasks and projects, and manage emotions (6). The definition of executive function deficit describes the symptoms of ADHD very closely because it is a main contributor to the disorder. Executive function issues also arise with menopause because of the loss of estrogen and its effects on dopamine.
A study by Epperson et al on the effects of lisdexamfetamine (LDX) in 32 perimenopausal and early postmenopausal women experiencing mid-life onset executive function difficulties revealed a significantly positive effect of LDX over placebo. LDX, also known as Vyvanse®, is a stimulant medication often used in the treatment of ADHD in adults and children. Stimulant medication enhances the dopaminergic system and improves executive function. It is important to note that none of the women in this study were diagnosed with ADHD prior to the onset of perimenopause and menopause (7).
Neurosteroids, neurotransmitters and brain function
Fluctuations in sex hormones affect the central nervous system and influence various brain areas that regulate mood, behavior, and cognitive abilities. Sex hormones are part of the larger category of steroid hormones. Steroid hormones with activity in the nervous system are called neurosteroids. Steroid hormones are mostly synthesized peripherally in the ovaries, adrenals, and adipose tissue and enter the nervous system by crossing the blood-brain-barrier. Neurosteroids can also be produced in the central and peripheral nervous systems by neurons and glial cells (5).
Neurosteroids participate in the regulation of neurotransmitters and neuronal excitability at the synaptic level. Neurotransmitters are signaling molecules that carry messages from one neuron to another. Dopamine is one of the main neurotransmitters responsible for focus and attention. Alterations in the dopamine signaling system are prevalent in ADHD and in menopause-related cognitive decline (8).
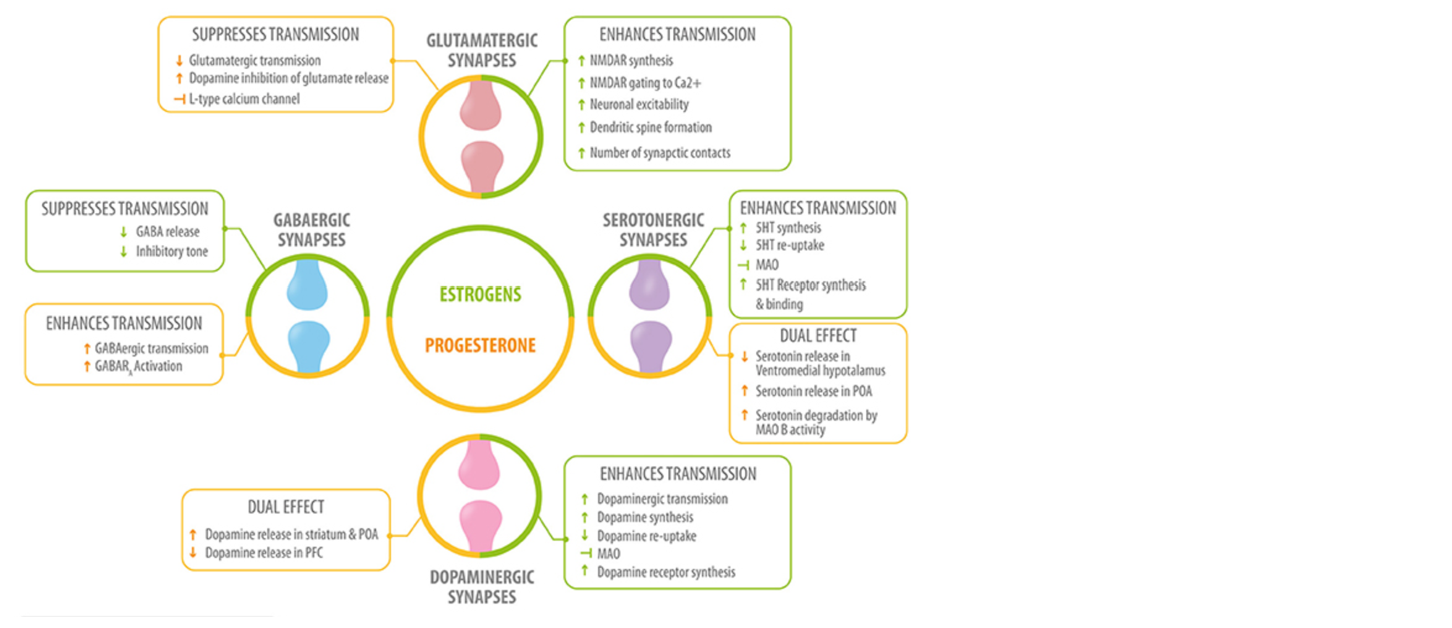
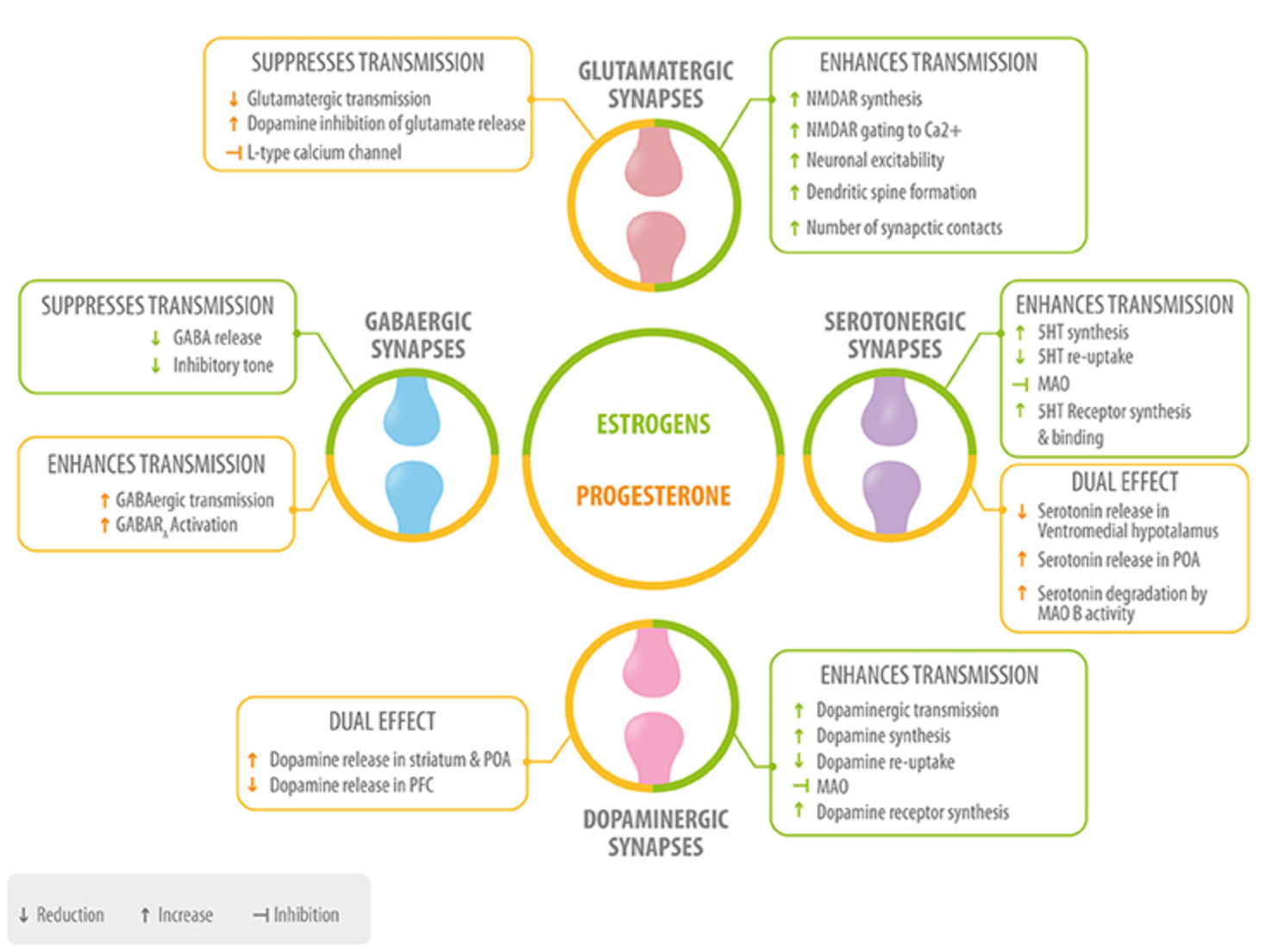
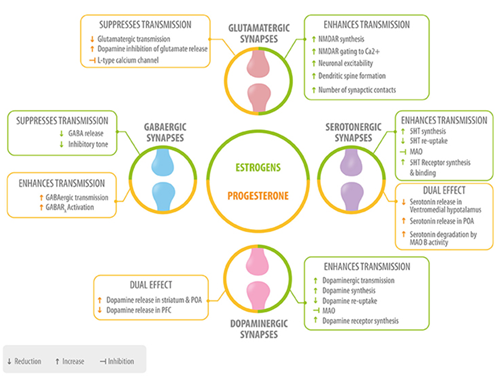
Fig 1. Role of neurosteroids in the modulation of the four main neurotransmitters. Estrogen (green) and progesterone (yellow) interact with GABAergic, glutamatergic, serotonergic, and dopaminergic synapses at different levels: neurotransmitter synthesis, release, degradation, and neurotransmitter receptor synthesis, activation or inhibition 5HT, serotonin; MAO, monoamino oxidase; preoptic area; PFC, prefrontal cortex. Del Rio JP, Alliende MI, Molina N, et al. Steroid hormones and their action in women’s brains: the importance of hormonal balance. Front Public Health. 2018;6:141. Creative Commons licensing.
Dopamine and ADHD
ADHD is viewed as a heritable neuropsychiatric condition linked to pathogenesis of brain dopamine. Molecular genetic studies have identified several genes that may mediate susceptibility to ADHD. The consensus in the literature on ADHD points to a dysfunction in the dopamine system that mediates the brain reward cascade. Blum et al see ADHD as part of a larger umbrella condition referred to as reward deficiency syndrome in which there is a disruption of the normal cascade of neurotransmitters that stimulate the reward centers of the brain (3). This may be why the decline in executive function in menopause is aptly described by the symptoms of ADHD.
The genetic trait that predisposes to ADHD is due in part to the D2 dopamine receptor (DRD2) A1 allele that prevents the expression of the normal production of dopamine receptors in brain reward sites. The brain lacks sufficient numbers of dopamine receptor sites to receive a normal amount of dopamine. This ultimately results in a reduction in dopamine production in the reward centers of the brain (3).
While the DRD2 gene may play a significant role in ADHD predisposition, it must be tied to a certain subset of additional genes for the clinical expression of ADHD (3). Additional genetic traits associated with ADHD include variants of the dopamine receptor D4 gene that influences the post-synaptic action of dopamine and DAT1, which is a dopamine transporter gene that mediates the reuptake of dopamine from the neural synapse (9). Stimulant medications commonly used to treat ADHD enhance dopamine signaling and improve the ability to focus.
Estrogen and dopamine
Through various signaling mechanisms, estrogen interacts with neurotransmitters that are highly involved in cognition and mood. Of the neurotransmitters that estrogen is involved with, the dopaminergic system is the most pronounced (10). Estradiol has been shown to impact working memory by enhancing dopamine activity and slowing reuptake (8).
Estradiol has a dopamine agonist-like effect on behavioral and neural processes that promote dopamine production and signaling. Certain estrogen metabolites (catechol estrogens) can also inhibit enzymes (catechol-o-methyltransferase) that inactivate dopamine. This is particularly true for dopamine in the pre-frontal cortex (PFC), which allows for the increased availability and stimulation of this powerful neurotransmitter (8). In short – increased estrogen and estrogen metabolites promote dopamine activity. If estrogen and its metabolites are low, the availability and action of dopamine in the PFC is also reduced.
Another mechanism by which estrogen influences dopamine levels and activity is through the DAT1 responsible for the reuptake of dopamine that ultimately terminates dopaminergic transmission. Estrogen inhibits DAT1 activity, which decreases uptake and increases the time for dopamine to exert its effect within the synaptic space (11).
Estrogen levels and cognitive abilities through the lifespan
Symptoms of ADHD appear differently in females than in males and cognitive abilities are affected by fluctuations in estrogen throughout the lifespan. As a woman enters perimenopause and menopause, cognitive complaints are common as hormones, particularly estradiol, are declining. The impact of declining estrogen on the dopaminergic system is a key component in the symptoms related to cognitive dysfunction and can present very similarly to ADHD. Estrogen clearly has neuroprotective effects over a woman’s lifetime as evidenced by studies reporting that prolonged exposure to estrogen results in better cognitive outcomes later in life (12).
In the brain, estradiol has neuroprotective effects that include modulation of neuropeptides and neurotransmitters, reduced cell apoptosis, modulation of neuronal growth and synaptic plasticity, support of mitochondrial activity, antioxidant properties, increased vasodilation and cerebral blood flow, regulation of brain glucose metabolism, reduced inflammation, and decreased formation of beta-amyloid, which is associated with the development of Alzheimer’s disease (12).
As women enter menopause, the timing of hormone replacement therapy is crucial for preserving cognitive function. The neuroprotective effects of estrogen are optimized early before deterioration within the brain and nervous system has occurred. This is referred to as the “healthy cell bias” (13). The sooner hormones are started, the better the outcome.
Between 1995 and 2006, the Cache County population-based study analyzed the records of 1,768 women who had provided a detailed history on the age at menopause and the use of hormone therapy. The study revealed that the women who used any type of hormone therapy within five years of menopause had a 30% reduced risk of Alzheimer’s disease. In contrast, women who started hormone therapy five years beyond the age of menopause showed increased rates of Alzheimer’s disease (14). As a woman’s neurological status progresses from healthy to unhealthy, the benefits of estrogen therapy decrease. If neurons are healthy at the time of estrogen exposure, their response to estrogen is beneficial for both neurological function and survival. In contrast, if neurological health is compromised, estrogen exposure over time exacerbates neurological demise (13).


Testing sex hormones, cortisol, and neurotransmitters
ZRT Laboratory offers a number of tests to measure sex hormones, cortisol, and neurotransmitters. As a young woman or a woman enters the middle phase of life, testing can provide valuable information regarding hormonal status, the stress response, and the balance between several neurotransmitters and metabolites.
Managing symptoms of ADHD or cognitive dysfunction related to menopause can create additional stress that may be revealed in a salivary adrenal stress profile. In the premenopausal years, assessing sex hormones within the menstrual cycle can reveal imbalances between estrogen and progesterone that may be contributing to symptoms related to cognitive dysfunction. If hormone replacement therapy is a consideration, testing hormones while in perimenopause or menopause can confirm the need for hormone replacement.
Neurotransmitter testing through ZRT provides a measurement of 14 neurotransmitters and metabolites that provides insight regarding the source of deficiency or excess. The information provided by hormone, adrenal, and neurotransmitter testing can be used to determine where to begin in the process of addressing executive function issues and cognitive dysfunction throughout a woman’s lifespan.
References
- Quinn PO, Madhoo M. A review of attention-deficit/hyperactivity disorder in women and girls: uncovering this hidden diagnosis. Prim Care Companion CNS Disord. 2014;16(3):PCC.13r01596.
- Antoniou E, Rigas N, Orovou E, et al. ADHD symptoms in females of childhood, adolescent, reproductive and menopause period. Mater Sociomed. 2021;33(2):114-118.
- Blum K, Chen AL-C, Braverman ER, et al. Attention-deficit-hyperactivity disorder and reward deficiency syndrome. Neuropsychiatr Dis Treat. 2008;4(5):893-918.
- Roberts B, Eisenlohr-Moul T, Martel MM. Reproductive steroids and ADHD symptoms across the menstrual cycle. Psychoneuroendocrinology. 2018;88:105-114.
- Del Río JP, Alliende MI, Molina N, et al. Steroid hormones and their action in women’s brains: the importance of hormonal balance. Front Public Health. 2018;23;6:141.
- Belsky G. What is executive function? Understood for All, Inc. Accessed August 4, 2023.
- Epperson CN Shanmugan S, Kim DR, et al. New onset executive function difficulties at menopause: a possible role for lisdexamfetamine. Psychopharmacology (Berl.) 2015;32(16):3091-3100.
- Jacobs E, D’Esposito M. Estrogen shapes dopamine-dependent cognitive processes: implications for women’s health. J Neurosci. 2011;31(14):5286-5293.
- Turic D, Swanson J, Sonuga-Barke E. DRD4 and DAT1 in ADHD: functional neurobiology to pharmacogenetics. Pharmacogenomics Pers Med. 2010;3:61-78.
- Hwang WJ, Lee TY, Kim NS, et al. The role of estrogen receptors and their signaling across psychiatric disorders. Int J Mol Sci. 2020;22(1):373.
- Dluzen DE. Unconventional effects of estrogen uncovered. Trends Pharmacol Sci. 2005;26(10):485-487.
- Conde DM, Verdade RC, Valadares ALR, et al. Menopause and cognitive impairment: a narrative review of current knowledge. World J Psychiatry. 2021;11(8):412-428.
- Brinton RD. Investigative models for determining hormone therapy-induced outcomes in brain: evidence in support of a healthy cell bias of estrogen action. Ann NY Acad Sci. 2005;1052:57-74.
- Shao H, Breitner JCS, Whitmer RA, et al. Hormone therapy and Alzheimer disease dementia. Neurology. 2012;79(18):1846-1852.
